
Meet Datwyler at
PDA Miniverse
Datwyler at the Forefront of European Parenteral Drug Packaging Innovation
Focused Technical Dialogue for Pharmaceutical Experts
PDA Miniverse brings together a focused community of European pharmaceutical professionals for intensive technical dialogue on combination products, medical devices, and connected health. Datwyler participates to engage quality, manufacturing, and regulatory teams from across the EU on the practical science of container closure integrity, extractables and leachables management, and high-performance elastomer selection for sterile injectable applications. This is the kind of event where real problems get solved — and our team comes prepared with the data, the expertise, and the collaborative mindset to help you find the right path forward.
Presentation Details
Enabling Large Volume Subcutaneous Delivery: A Collaborative Approach among Suppliers to Accelerating Market Readiness
Meet the Speakers


Valerio Ravazzolo
Valerio Ravazzolo is Product Manager for the Cartridge Platform within the DCS Business Unit at Stevanato Group, where he focuses on the evolution of cartridge solutions from both a technical and market perspective.
He joined Stevanato Group in 2015, starting in Quality Assurance and later moving into a Technical Account Manager role. In this position, he supported key customers and gradually expanded his scope of responsibility.
Today, as Product Manager, he brings together this experience to support product strategy, development, and long‑term platform growth.
Valerio holds a master's degree in mechanical engineering from the University of Padua.
Greg Moakes
Greg Moakes is Executive Vice-President of Business Development for LTS Lohmann Device Technologies. Dr Moakes holds a PhD in Chemistry from the Georgia Institute of Technology (Atlanta, GA, US) and an MBA from the Southern Methodist University (Dallas, TX, US). His career had been focused on drug delivery, first specialising in solid-implant dosage formulations and more recently high-volume subcutaneous delivery via on-body delivery systems.
What We're Showcasing at PDA Miniverse
Explore the solutions and technologies most relevant to this event.

Presentation: Enabling Large-Volume Subcutaneous Delivery — A Collaborative Approach Among Suppliers
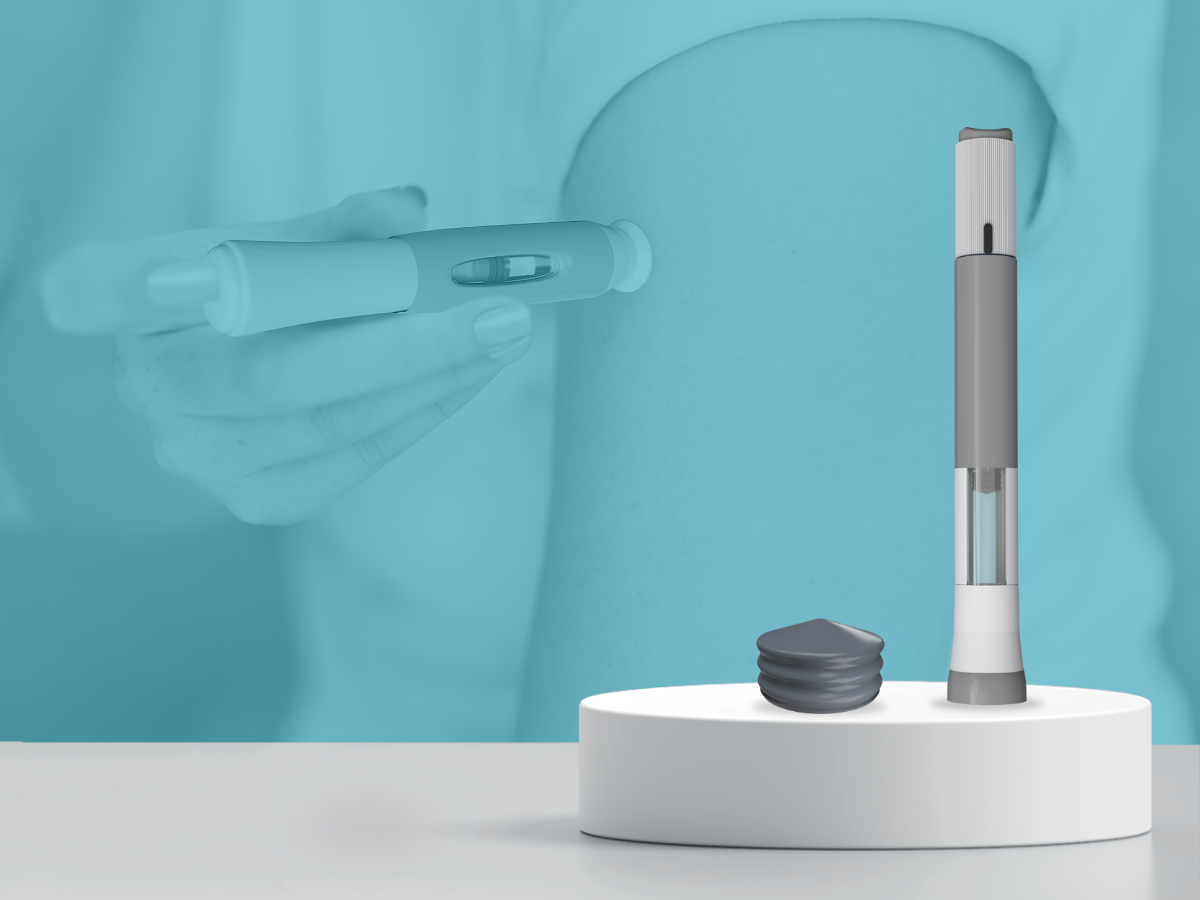
First-to-Market 20ml Universal Plunger for Large-Volume Injectables

Drug Delivery Solutions for Home-Based & Patient-Centric Care
Continue Exploring
Access additional materials and information related to this event and Datwyler’s solutions.
