Featured Resource
The New Subcutaneous (SC) Landscape: Adapting Drug Delivery for Biologics Administration at Home
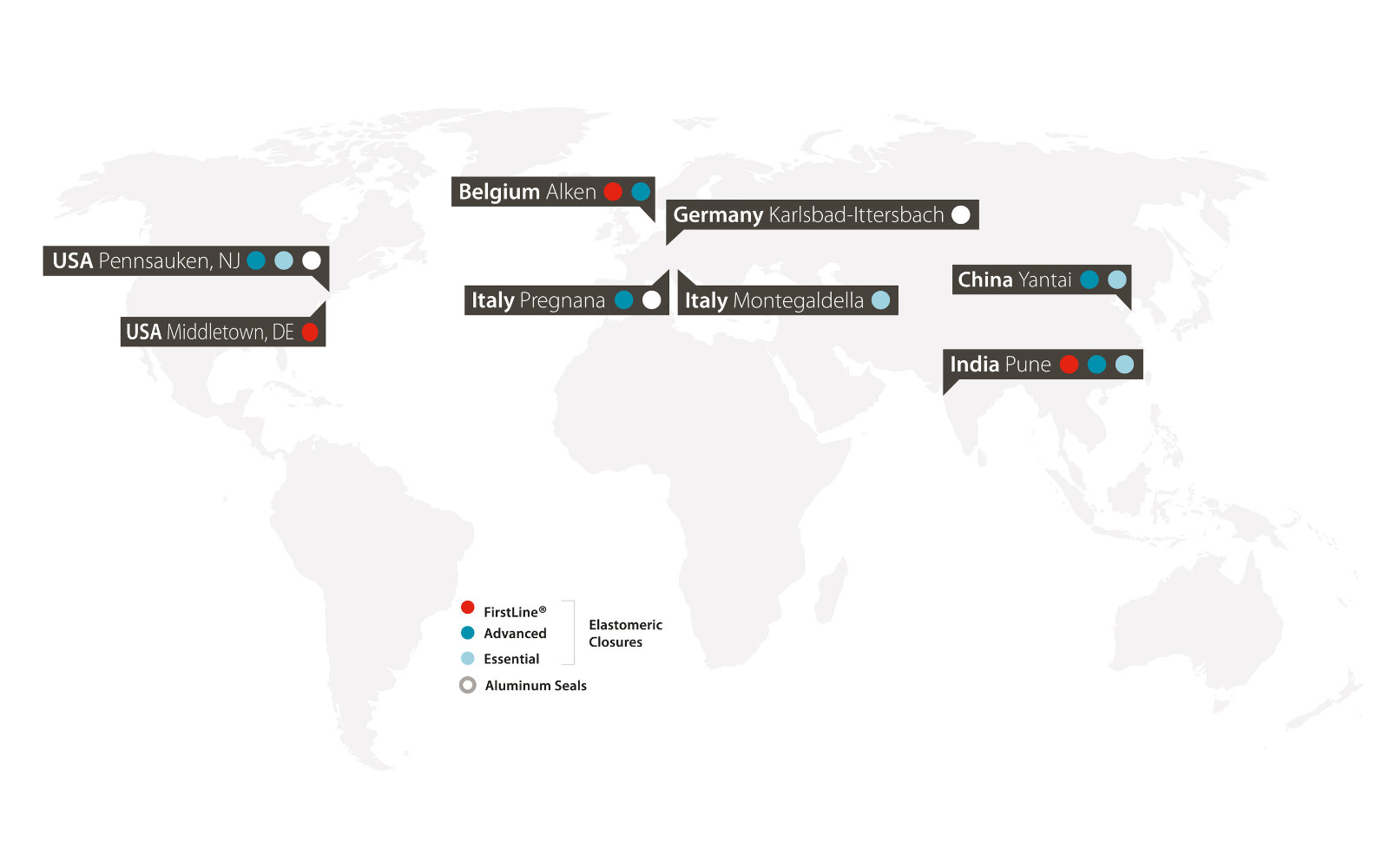
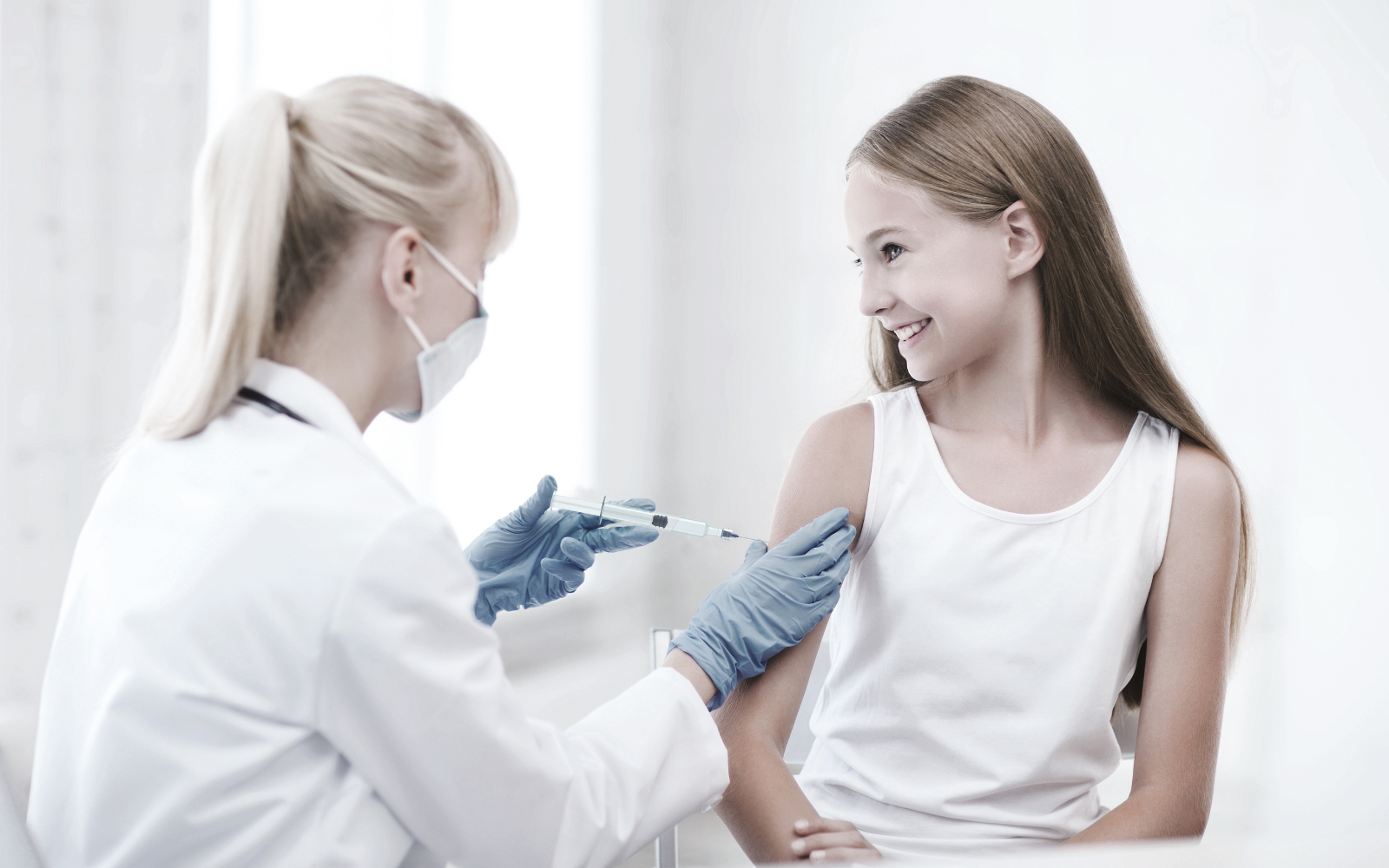
This on-demand webinar examines the structural shift toward large-volume subcutaneous self-administration for biologics — and presents the first concrete evidence of a complete 20 mL on-body injection system, featuring components from Datwyler, Stevanato Group, and LTS Device Technologies, using V9621 NeoFlex™ plungers in the EZ-fill® 20 mL cartridge with the Sorrel™ wearable platform — with CCI, break-loose force, gliding force, dose accuracy, delivery time, and hold-up volume data.

.png)
.png)





.png)



.png)




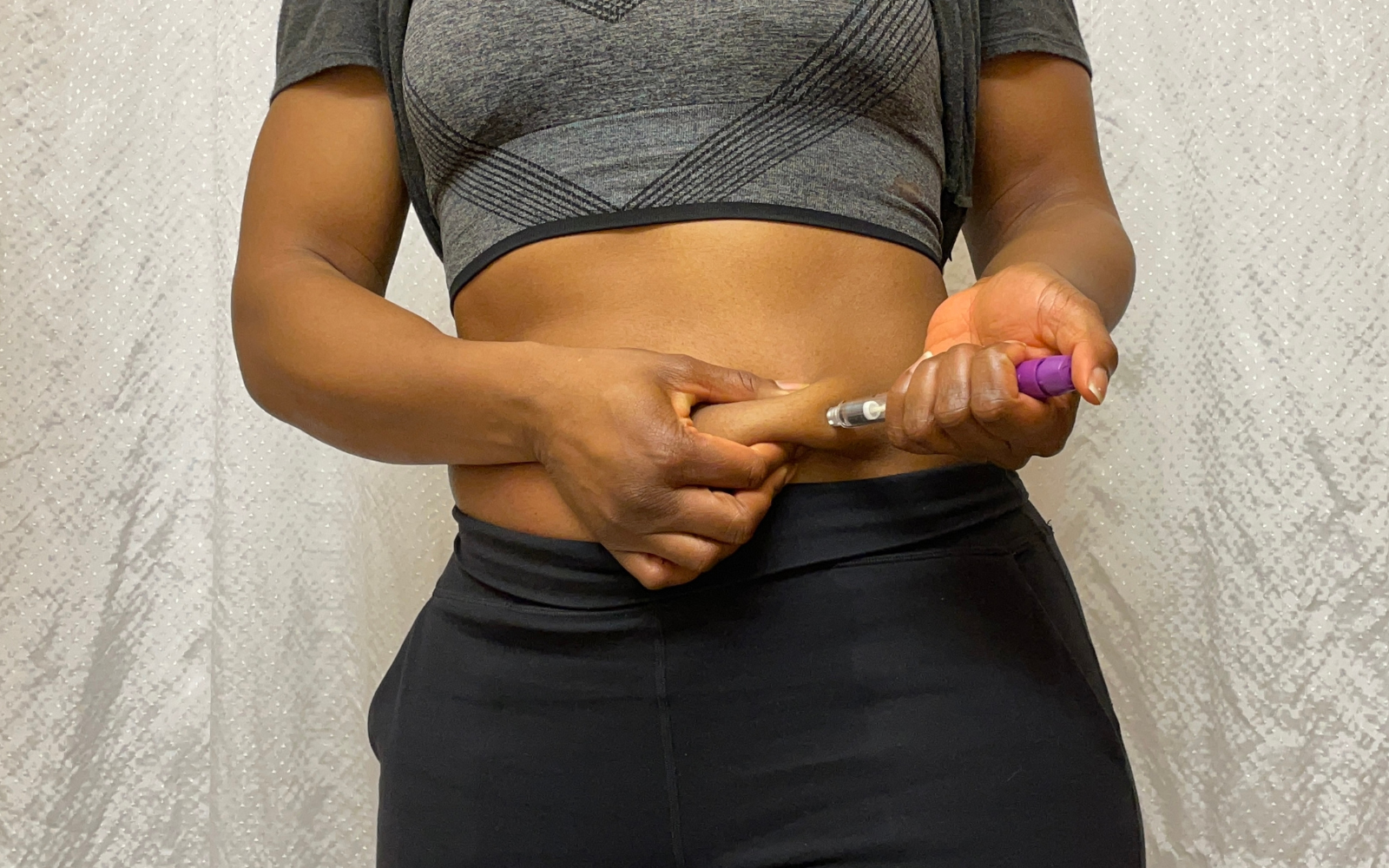


.png)












.png)




.png)