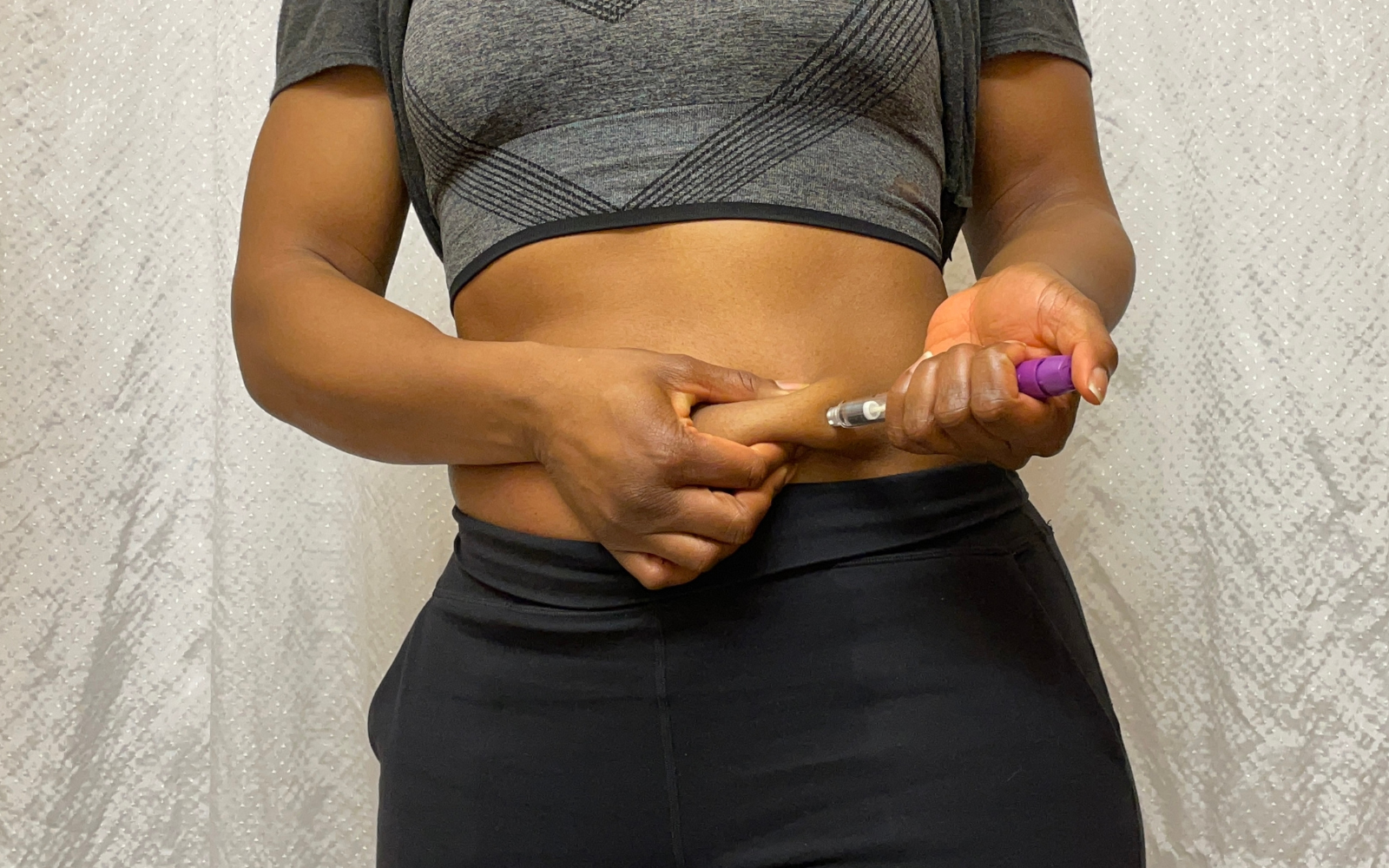Guide
GLP-1 Assurance Checklist: Compatibility, Compliance, and Confidence
From FM457 compound purity and NeoFlex™ plunger gliding performance to EU GMP Annex 1-compliant RTP bag configurations and dual sourcing continuity planning, this checklist provides the technical and operational criteria to select GLP-1 primary packaging components with confidence.
Status
Published
Format
Guide
Language
English
Access
Registration Required
Resource Overview
About this Checklist
GLP-1 receptor agonist therapies — increasingly used for obesity and type 2 diabetes in self-administered pen and auto-injector formats — place exceptional demands on elastomer primary packaging components. Chemical sensitivity to oxidation, aggregation, and drug-component interaction requires ultra-low extractables compounds such as FM457 and inert fluoropolymer coatings such as NeoFlex™, with PFAS regulatory readiness built into material selection from the outset. Functional performance criteria for plungers span break-loose and gliding forces at varying temperatures, plunger movement under air transport vacuum conditions, and container closure integrity (CCI) for full shelf-life sterility — while combination aluminum-rubber seals (combiseals) must demonstrate multi-piercing resealability of 50–100 piercings and fragmentation resistance after needle penetration. Device compatibility requires validation against leading pen and auto-injector platforms, with RTU and RTP bag formats supporting EU GMP Annex 1-compliant aseptic processing. Regulatory assurance is anchored by FirstLine® Grade C cleanroom manufacturing, ISO 15378 production standards, and DMF filing support across the US, EU, China, Japan, and India. Supply chain resilience and lifecycle management complete the framework, including a two-by-two matrix strategy for qualifying multiple interchangeable components to protect against future regulatory or format transitions.
Access This Resource
Download the Guide
Continue to access the full resource.
Access the Guide
Quick access in under 30 seconds.